View Compound With Trigonal Planar Geometry Background
View Compound With Trigonal Planar Geometry Background. In this case bf3 has. G.) and molecular geometry is bh3.
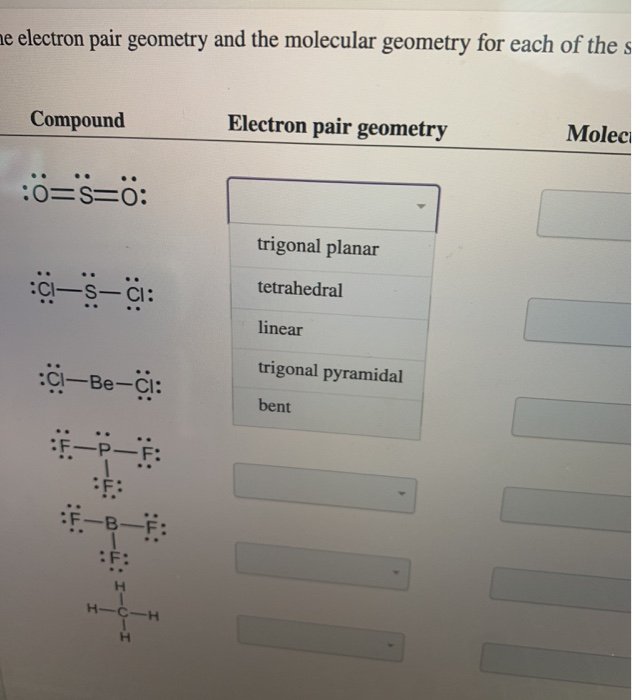
Trigonal planar is a molecular shape that results when there are three bonds and no lone pairs around the central atom in the molecule.
The smallest possible repeating unit is called a unit cell. Examples of molecules with trigonal planar geometry include boron trifluoride (bf3), formaldehyde (h2co), phosgene (cocl2), and sulfur in chemistry, trigonal planar is a molecular geometry model with one atom at the center and three atoms at the corners of an equilateral triangle, called peripheral. This molecule is made up of 3 equally spaced sp 2 hybrid orbitals arranged at 120 o angles. Carbon dioxide cannot be the answer b/c the c is surrounded by only two other atoms.
Posting Komentar untuk "View Compound With Trigonal Planar Geometry Background"